
Carbon is more electronegative than Hydrogen. If there are two atoms and both of them are not identical, the magnitude of the vector will not be zero, and the bond will not be polar. When there are complicated molecules, it becomes necessary to consider the expectations of molecular polarities that born from the combination of all the single bond polarities. When there is different electronegativity in the atoms, there are cases of polarities. You can also memorize that AX2 is a linear molecular geometry. It is a very simple molecule, and you can see that it is linear with the bond angles of degrees. If you want to confirm the status, then look at up at the table. Here, A would be the central carbon, X would be the number of atoms attached to that central carbon In HCN, there are two - Hydrogen and Nitrogen and N would be the number of lone pair electrons or nonbonding electron pairs. A, X, and Notation theory can also be used to make sure about the right molecular geometry. Electrons around the Carbon atom are involved in chemical bonds. Lewis structure is used to predict the overall geometry of a molecule or ion and the number of electrons surrounding a central atom. Molecular geometry is known as the specific three-dimensional arrangements of atoms in molecules. It should be called like - H single bond C triple bond N with a lone pair of electrons sitting on the end.
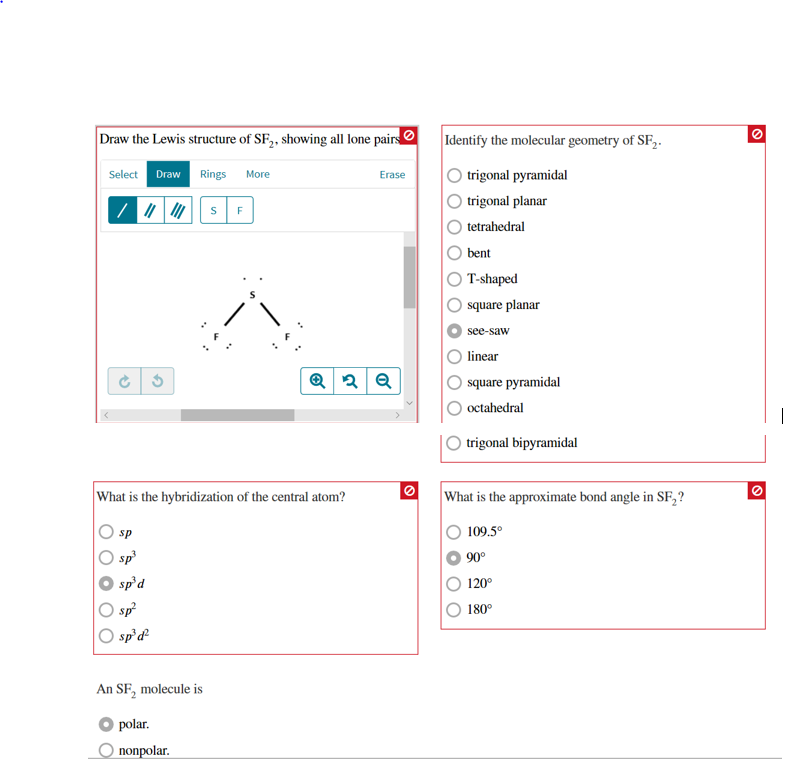
#SF2 MOLECULAR GEOMETRY FULL#
Once the least electronegative atom in the center fills the outer atoms, move outer electron pairs to the center until they have a full octet. This is how Lewis dot structure of Hydrogen Cyanide goes! Bonding-polar vs nonpolar molecules -keeping it simple So assuming that the valence shell electron pairs are repelling each other, there will be a Hydrogen on one side and the Nitrogen on the other hand of the central atom Carbon. According to VSEPR theory, the atoms are going to spread out as far as possible while still staying connected to that central carbon. Understanding of HCL can be beneficial to you if you are a chemistry freak. HCN Lewis Structure, Molecular Geometry, Shape, and Polarity
#SF2 MOLECULAR GEOMETRY SKIN#
It can affect the body by ingestion, inhalation, skin contact, or eye contact. One can release it as a liquid spray or a gas into the indoor air or outdoor air or can be used to contaminate food or water. There are few other names like Formonitrile, Hydrocyanic acid, and Prussic acid. HCL can also be used as a solution in water. It has a distinctive bitter almond odor, and some also say that it smells like some old sneakers smell. HCL interferes with the regular use of Oxygen by the organs of the body. It is colorless gas above 78 degrees Fahrenheit. Frequently Asked Questions What scientific concept do you need to know in order to solve this problem? What is the difficulty of this problem?If not in the liquid form, it can be in the gas form. Which statement best summarizes the difference between ionic and molecular compounds?Ī Molecular compounds contain highly directional covalent bonds, If the atoms that share electrons have an unequal attraction for the electrons, the bond is called? Which of the following molecules does not have a permanent dipole moment? What is the percent ionic character of the HI bond? Molecules can be polar because What is a chemical bond? Give the difference between an ionic bond and a covalent bond. Learn this topic by watching Dipole Moment Concept Videos.
#SF2 MOLECULAR GEOMETRY FREE#
Join thousands of students and gain free access to 46 hours of Chemistry videos that follow the topics your textbook covers.Īnalytical Chemistry Video Lessons.
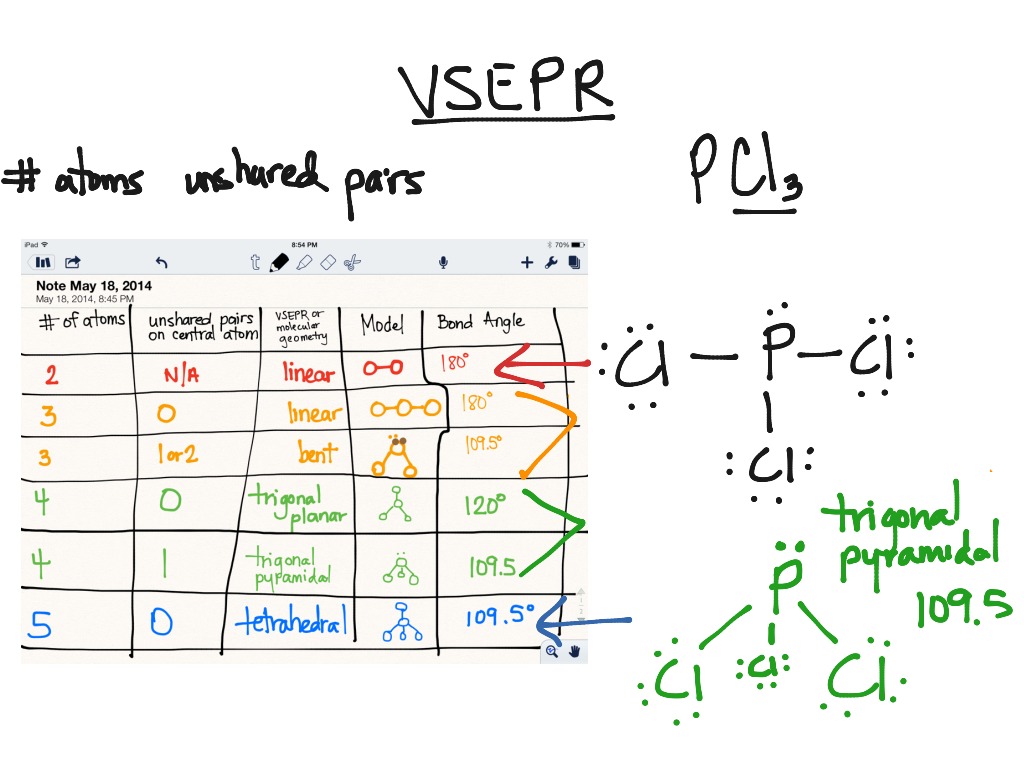
If you forgot your password, you can reset it. You can follow their steps in the video explanation above. Our expert Chemistry tutor, Sabrina took 2 minutes and 6 seconds to solve this problem. Or if you need more Dipole Moment practice, you can also practice Dipole Moment practice problems. You can view video lessons to learn Dipole Moment. What scientific concept do you need to know in order to solve this problem? Our tutors have indicated that to solve this problem you will need to apply the Dipole Moment concept.
